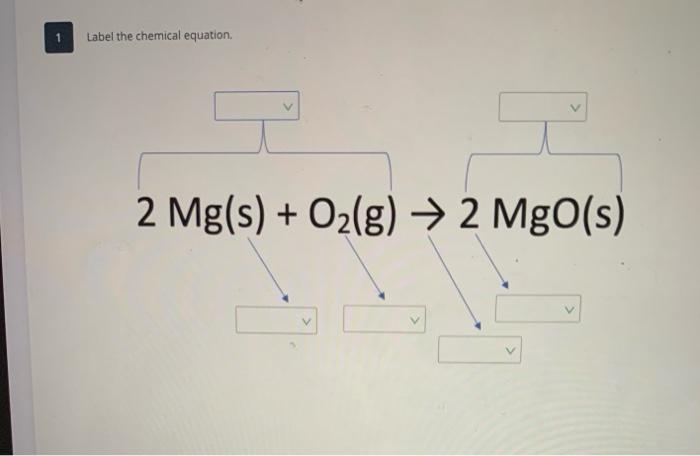
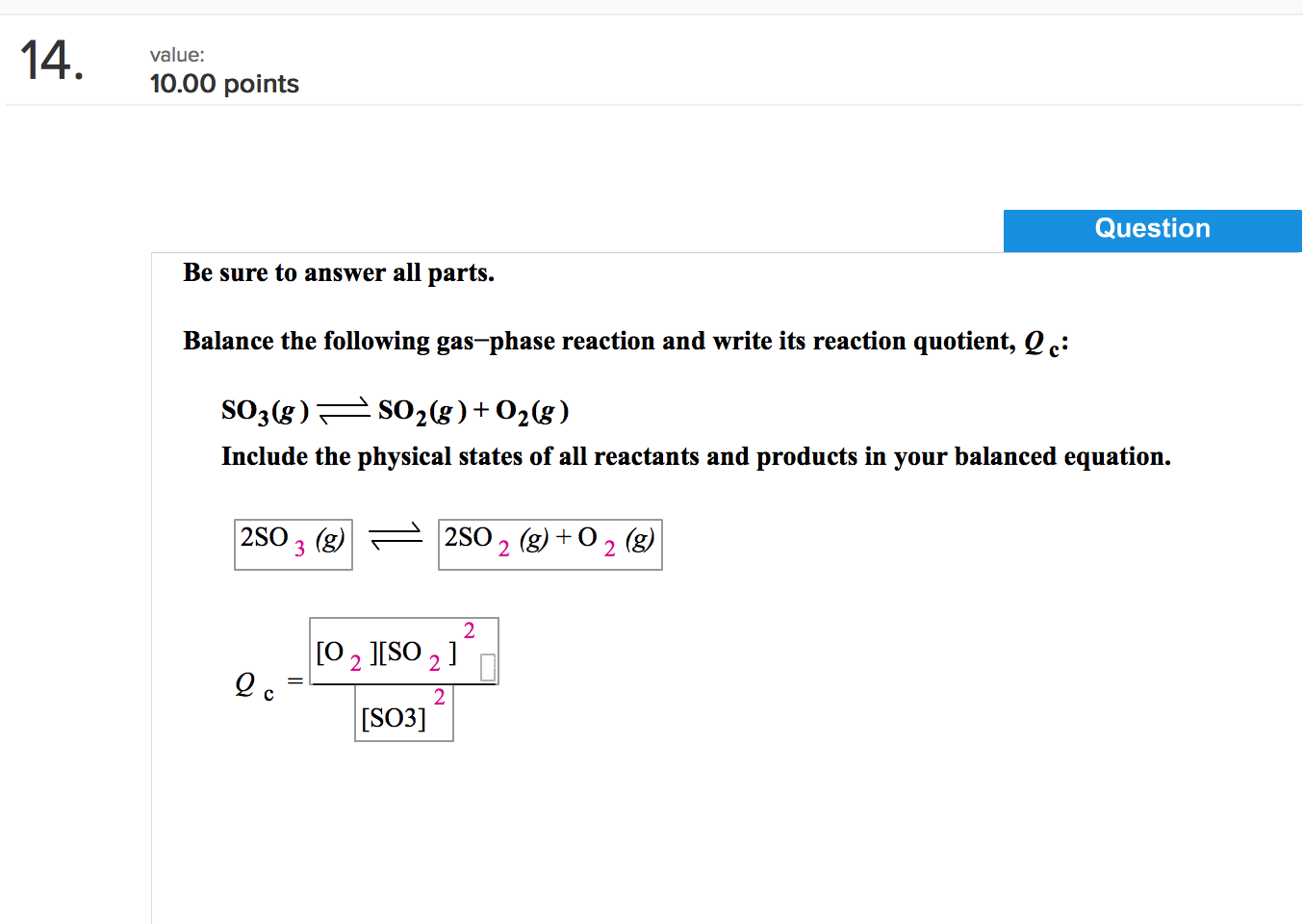
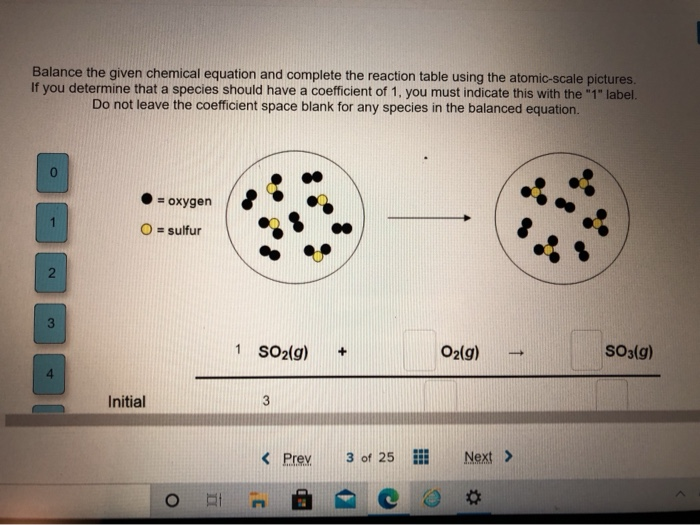
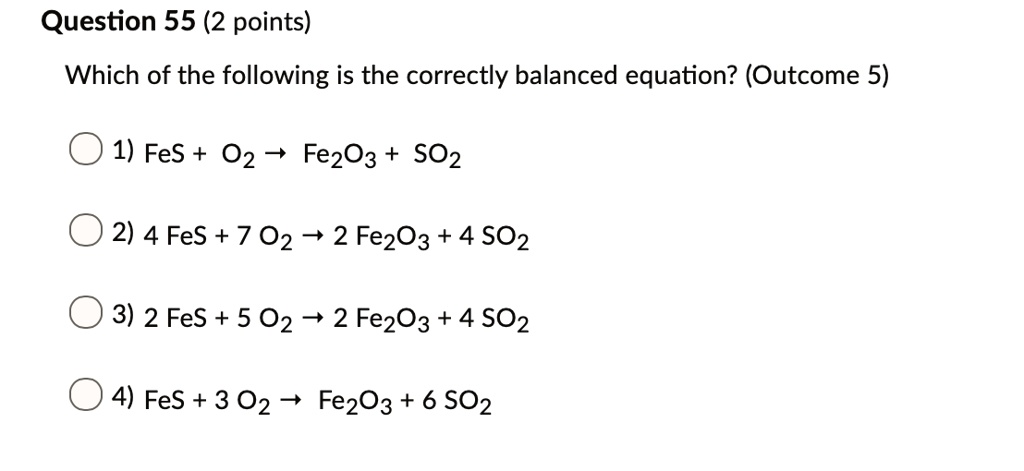PPT - 1. Write a sentence that describes this chemical reaction. PowerPoint Presentation - ID:3870081
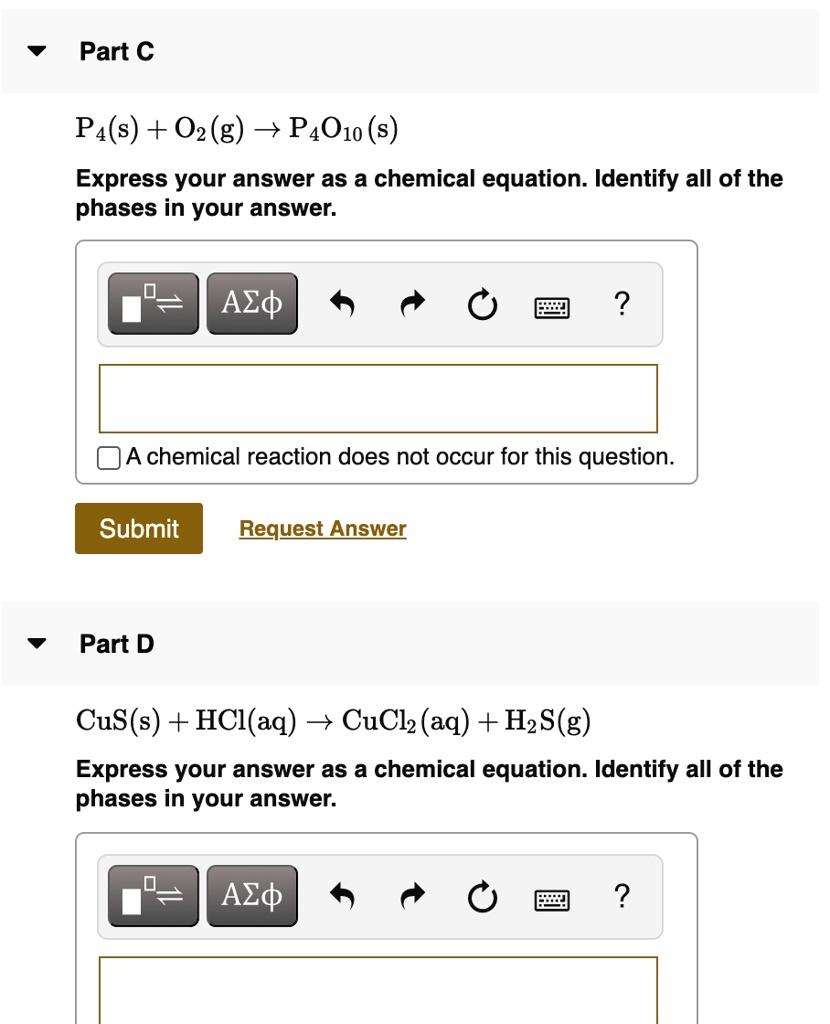
SOLVED:Part € Pa(s) + O2(g) - P4O1o (s) Express your answer as a chemical equation. Identify all of the phases in your answer: Azd A chemical reaction does not occur for this
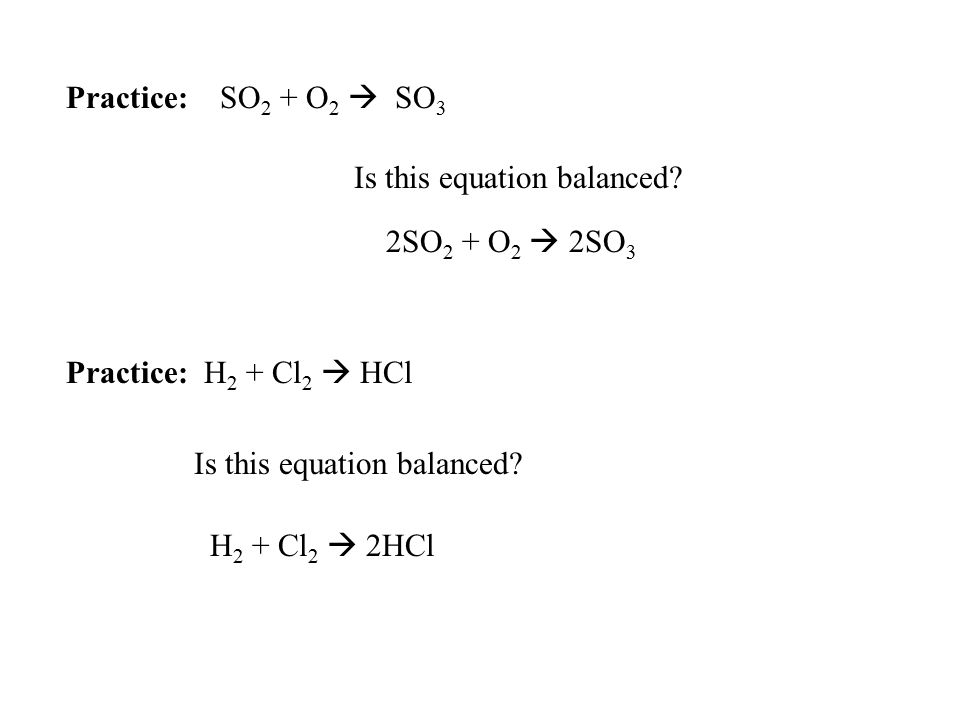
Chemical Equations Chemical Reaction: Interaction between substances that results in one or more new substances being produced Example: hydrogen + oxygen. - ppt video online download