Applied Sciences | Free Full-Text | Hydrogen Storage: Thermodynamic Analysis of Alkyl-Quinolines and Alkyl-Pyridines as Potential Liquid Organic Hydrogen Carriers (LOHC) | HTML

J. Am. Chem. Soc. on Twitter: "Out now! A convenient new method to prepare complex N-substituted piperidine ring systems which is robust and diastereoselective. Check it out! #MerckChemistry https://t.co/FY1VMXOqfr https://t.co/Wb9PhcUMky" / Twitter

Fentanyl-related compounds and derivatives: current status and future prospects for pharmaceutical applications. - Abstract - Europe PMC
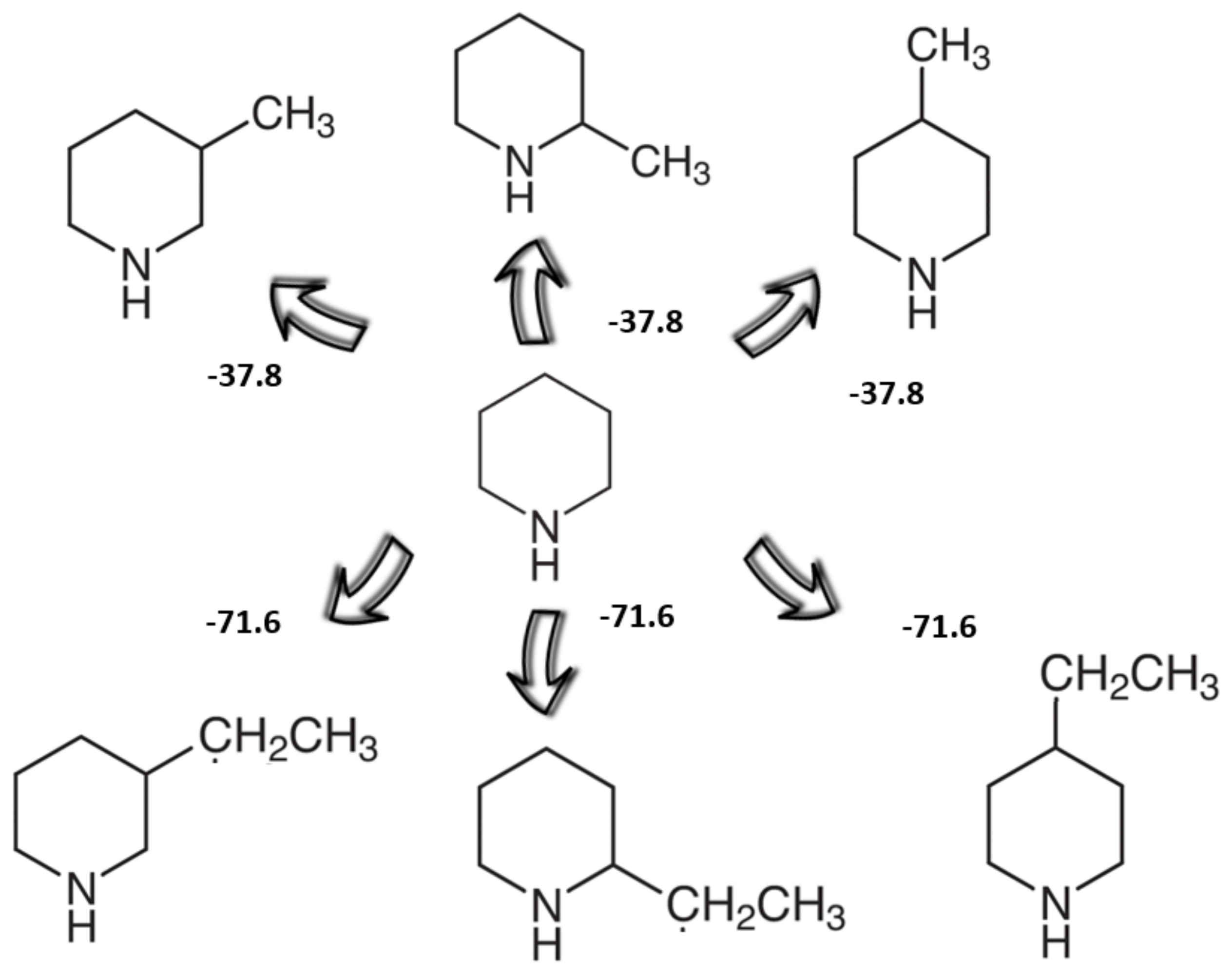
Welcome to Chem Zipper.com......: Pyridine is almost 1 million times less basic than piperidine? Why?

Some compounds containing a piperidine ring in their structures. (A)... | Download Scientific Diagram

Benzyne‐Induced Ring Opening Reactions of DABCO: Synthesis of 1,4‐Disubstituted Piperazines and Piperidines - Seo - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

What is the expected number of rings obtained in the following ring formation process? - Chemistry Stack Exchange

Plausible mechanism of 3,5-dispirosubstituted piperidine ring formation | Download Scientific Diagram
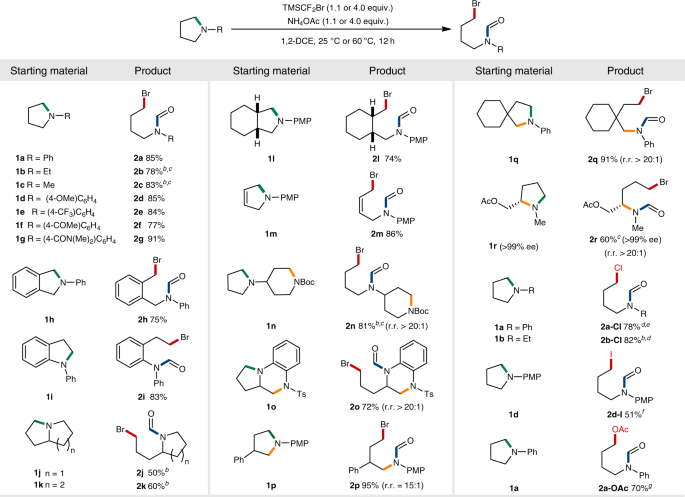
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications

Recent progress toward the asymmetric synthesis of carbon-substituted piperazine pharmacophores and oxidative related heterocycles - RSC Medicinal Chemistry (RSC Publishing)
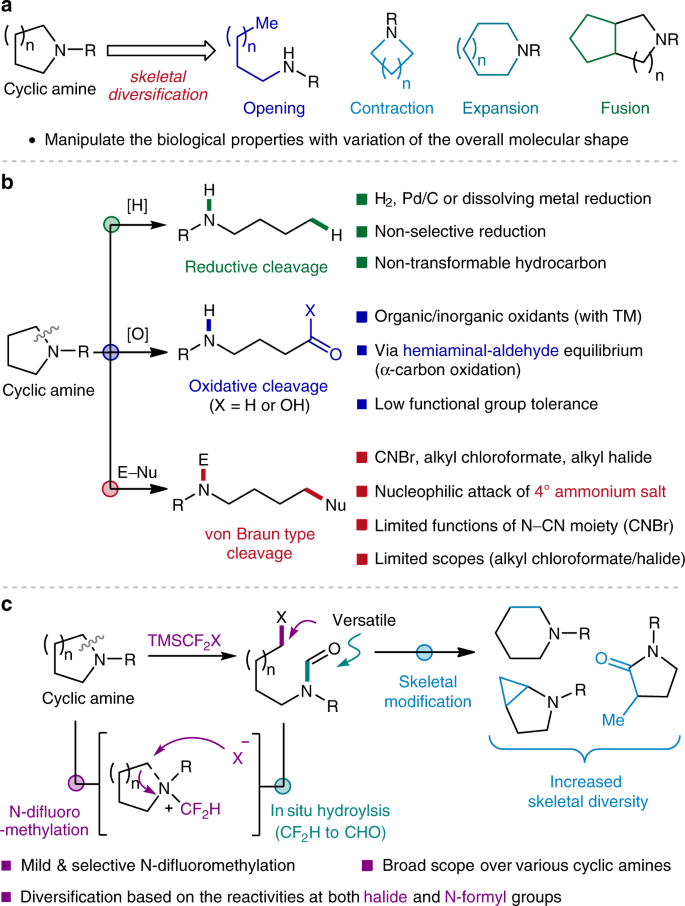
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications
/DB19273E6550AD06802585F9007F9729/$file/FP36451_structure.png)